40 multi step reaction energy diagram
E1 Reaction Mechanism and E1 Practice Problems - Chemistry Steps Step 1: Loss of he leaving group. The energy diagram of the E1 mechanism demonstrates the loss of the leaving group as the slow step with the higher activation energy barrier: The dotted lines in the transition state indicate a partially broken C-Br bond. The Br being the more electronegative element is partially negatively charged and the ... Rate-determining step - Wikipedia In a multistep reaction, the rate-determining step does not necessarily correspond to the highest Gibbs energy on the reaction coordinate diagram. [8] [6] If there is a reaction intermediate whose energy is lower than the initial reactants, then the activation energy needed to pass through any subsequent transition state depends on the Gibbs ...
Label the following multi-step reaction energy diagram. - Chegg Expert Answer. 99% (128 ratings) Transcribed image text: Label the following multi-step reaction energy diagram.

Multi step reaction energy diagram
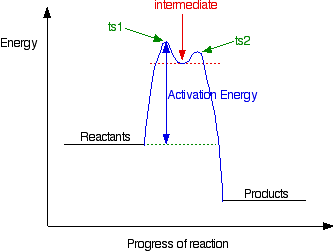
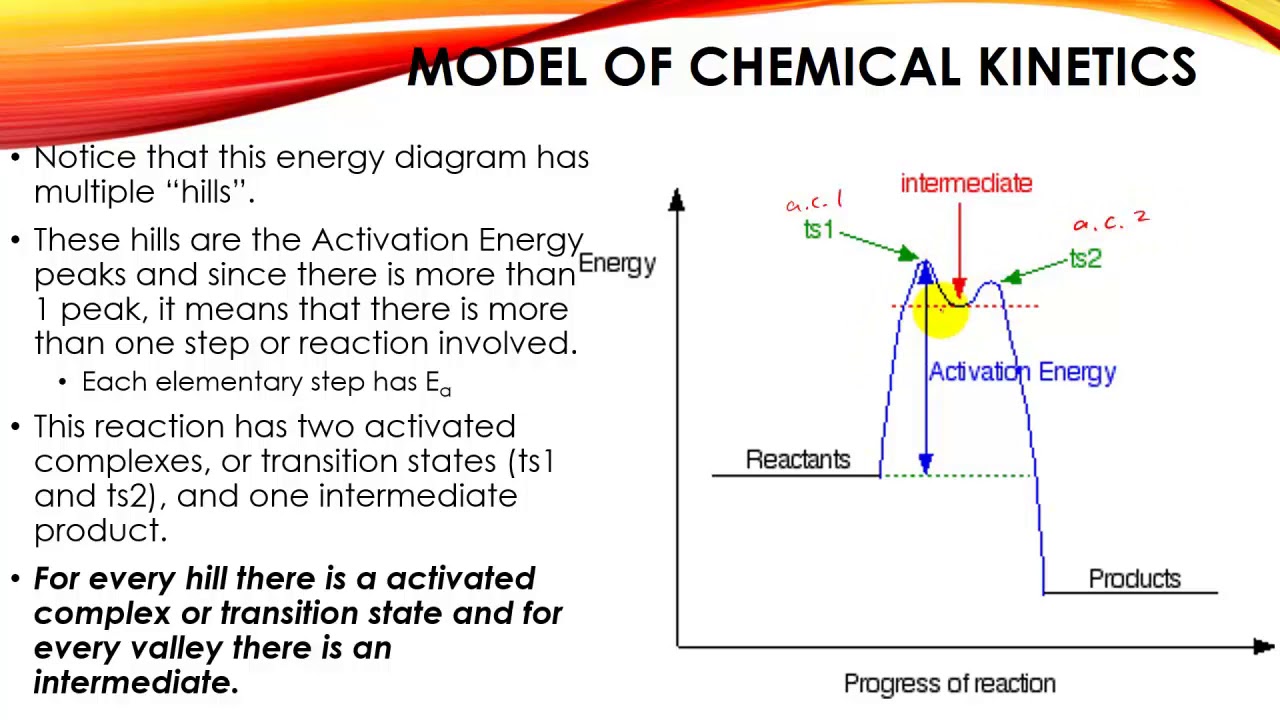
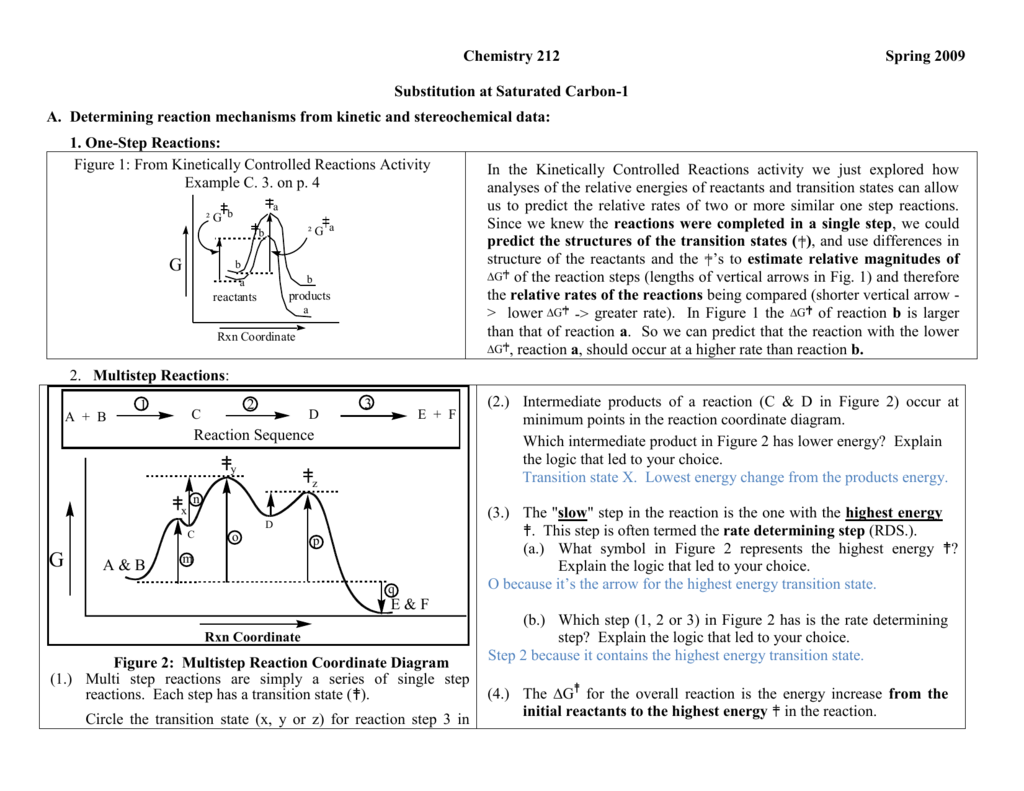
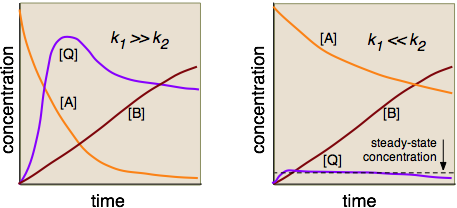
Reaction Mechanisms and Multistep Reactions - Chemistry LibreTexts Multi-step (Consecutive) Reactions Mechanisms in which one elementary step is followed by another are very common. A+ B → Q B+ Q → C A+ 2B → C (As must always be the case, the net reaction is just the sum of its elementary steps.) In this example, the species Q is an intermediate, usually an unstable or highly reactive species. Energy Diagrams of Reactions | Fiveable The energy diagrams below show what should be known for the test. Image Courtesy of Pinterest Before looking at the specifics of each, you should be aware of a few terms: PEreactants - The potential energy of the reactants PEproducts - The potential energy of the products Activation Energy - Energy necessary for the reaction to occur Mechanisms and Potential Energy Diagrams - Course Hero The potential energy diagram can illustrate the mechanism for a reaction by showing each elementary step of the reaction with distinct activation energy (see Figure below ). Figure 1. The potential energy diagram shows an activation energy peak for each of the elementary steps of the reaction.
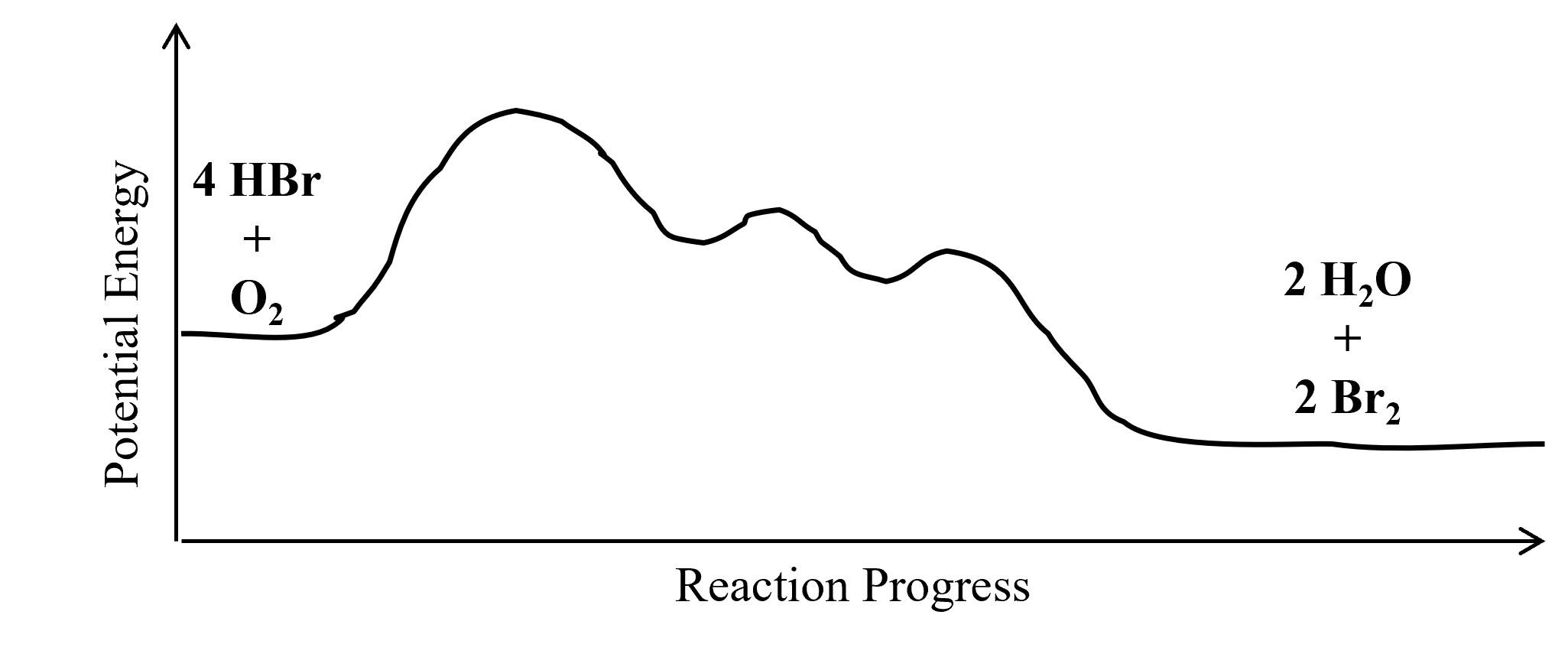
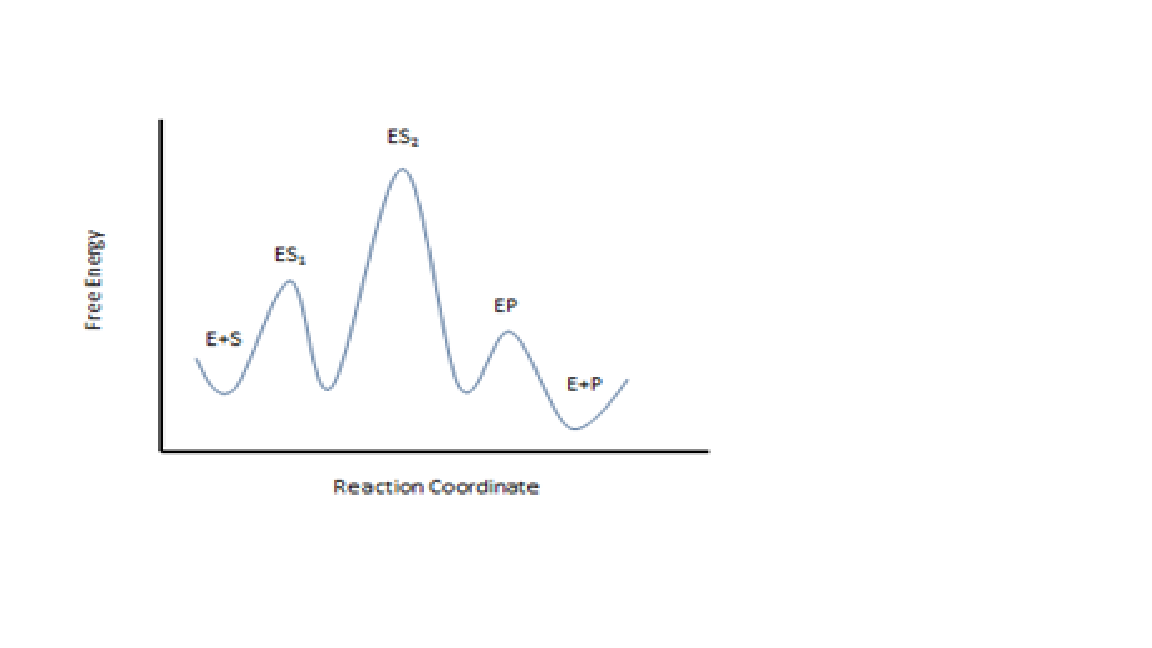
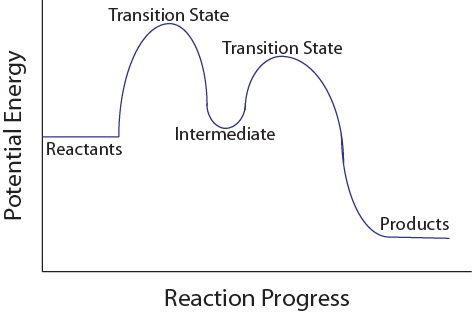
Multi step reaction energy diagram. Answered: Reaction progress Potential Energy | bartleby Q: The diagram below shows the energy profile for a reaction. D В E C A Reaction progress Species… D В E C A Reaction progress Species… A: transition state is a very short lived state of atoms ,in the transition state , Energy of atoms… Energy Diagrams of Two Step Reactions - YouTube Watch Complete videos @ Organic Chemistry 1 Multistep Reactions - Softschools.com The energy diagram of a two-step reaction is shown below. In the above reaction, a reactant goes through one elementary step with a lower activation energy (transition state 1) to form the intermediate. The intermediate then goes through a second step (transition state 2) with the highest energy barrier to form the product. Energy Diagram Module Series- Part Three: Intermediates and Rate ... Sometimes reactions are more complex than simply a transition state (Graph 3), which would represent a single step in the reaction mechanism. You will soon see most reactions proceed in a multistep fashion. In this case, reaction mechanisms often form lower energy and sometimes isolatable intermediates.
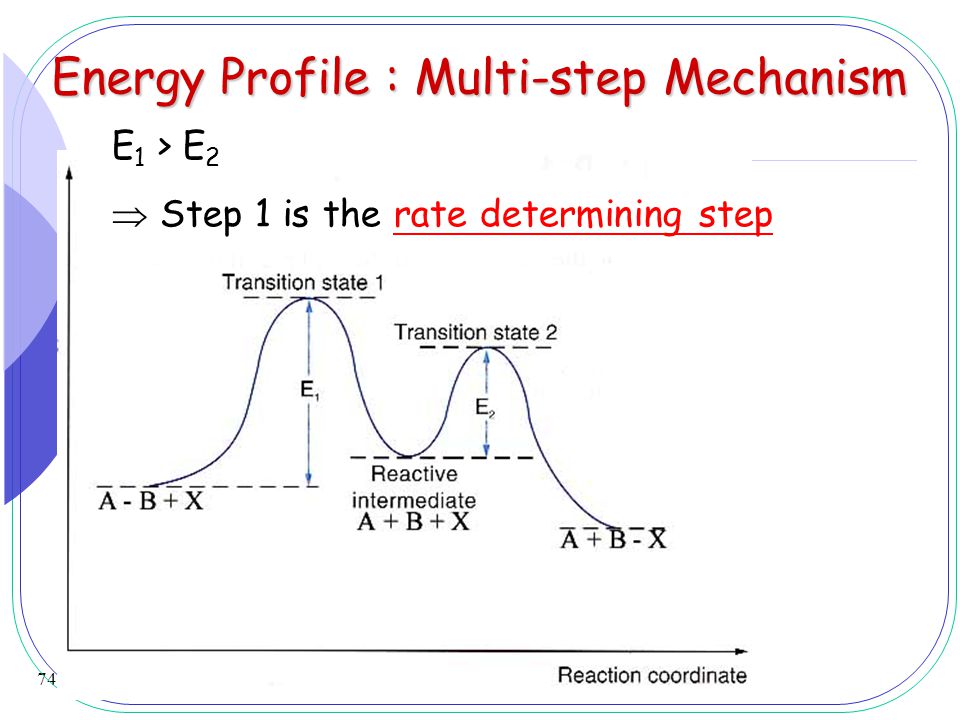
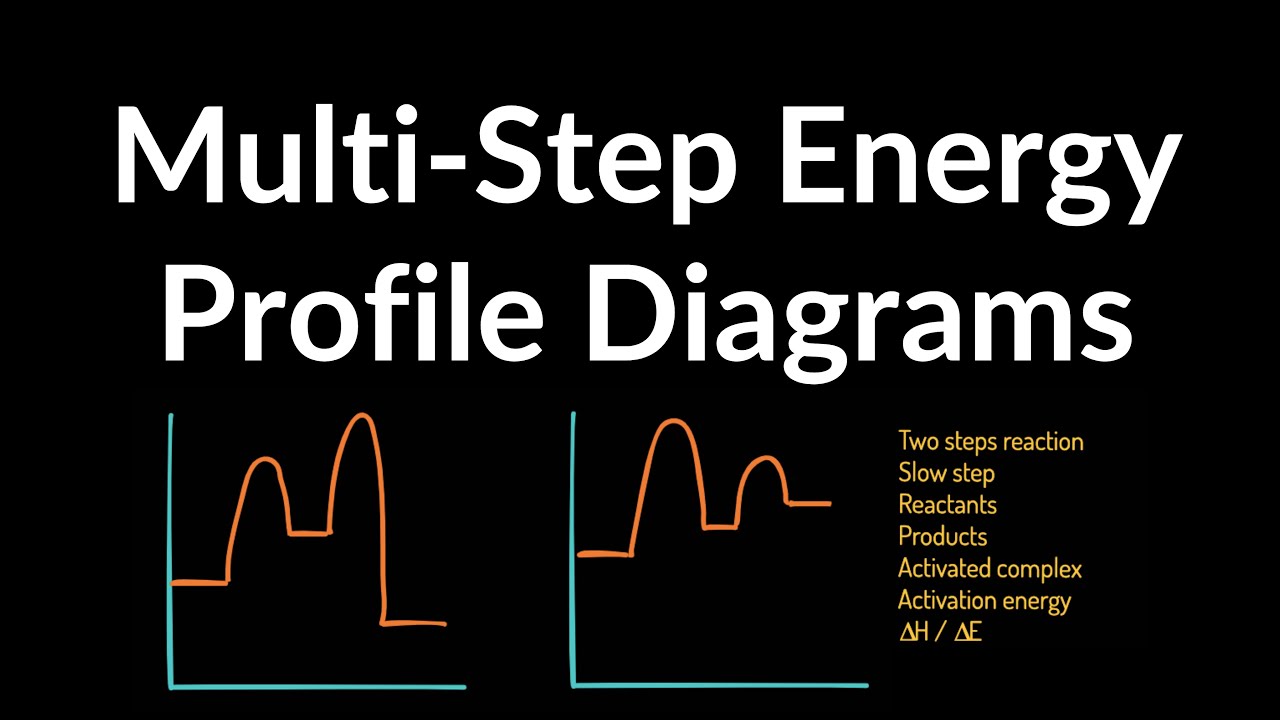
How to Draw Multi-Steps Energy Profile Diagrams: Reactant ... - YouTube We'll go over how to label the reactants, products, transition state, activated complex, activation energy, ∆H delta H, forward reaction, reverse reaction. We'll go over how the activation energy... 7.4: SN1 Reaction Mechanism, Energy Diagram and Stereochemistry Because S N 1 is a multiple-step reaction, so the diagram has multiple curves, with each step can be represented by one curve. Out of the three steps, the activation energy for step 1 is the highest, therefore step 1 is the slowest step, that is the rate-determining step. Figure 7.4a Energy diagram for SN1 reaction between (CH3)3CBr and H2O Ch 16 Pt 2 Smartbook Flashcards | Quizlet Multiple choice question. The energy threshold that the colliding particles must exceed in order to react. A reaction energy diagram plots potential ________ versus reaction _________ . energy, progress. Analyze the reaction energy diagram and match the values and their labels correctly. -y-axis: Potential energy. -x-axis: Reaction progress. Apparent Activation Energies in Complex Reaction Mechanisms: A Simple ... This simplicity provides deep insight into the connection between the reaction energy diagram and the apparent activation energy. We prove both this and the quantitative validity of this equation by analysis of numerous reaction mechanisms. ... (DRC) is a mathematical approach for analysing multi-step reaction mechanisms that has proven very ...
Energy Diagrams: Describing Chemical Reactions Draw an energy diagram for a two-step reaction that is exothermic overall, and consists of a fast but endothermic first step, and a slow but exothermic second step. Indicate DGrxn, as well as DG1* and DG2* for the first and second activation energies, respectively. Label the positions corresponding to the transition states with an asterisk. Reaction Mechanisms - Chem1 2 Multi-step (consecutive) reactions. Mechanisms in which one elementary step is followed by another are very common. step 1: A + B → Q. step 2: B + Q → C. net reaction: A + 2B → C. (As must always be the case, the net reaction is just the sum of its elementary steps.) In this example, the species Q is an intermediate, usually an unstable ... Chapter 6: Understanding Organic Reactions Flashcards | Quizlet Which of the following statements about a two-step reaction mechanism is true? A) The transition states are located at energy minima. B) Each step is characterized by its own value of DH° and Ea. C) The rate-determining step has the lower energy transition state. D) The reactive intermediate is located at an energy maximum. Difference Between Concerted and Stepwise Reactions The key difference between concerted and stepwise reactions is that concerted reactions are single-step reactions, whereas stepwise reactions are multistep reactions.. The terms concerted and stepwise reactions come under the field of physical chemistry where the rates of reactions are determined using the change in the reactant and product amounts with time.
Energy Diagram for a Two-Step Reaction Mechanism Step 1 has the higher transition energy state, thus it is the rate-determining step. In many reactions more than one step is involved in the formation of products. Step Two Exothermic because energy is released in forming B-C bond delta H is negative Products are lower than the starting materials
Reaction mechanism and rate law (article) | Khan Academy Summary. A reaction mechanism is the sequence of elementary steps by which a chemical reaction occurs. A reaction that occurs in two or more elementary steps is called a multistep or complex reaction. A reaction intermediate is a chemical species that is formed in one elementary step and consumed in a subsequent step.
energy diagrams chemistry - TeachersPayTeachers by. Doc Mathewson. $3.99. PPTX. This is a Power Point on the Reaction Mechanisms and Potential Energy Diagrams. Other topics include elementary steps, reaction intermediates, rate determining step and labelling enthalpy, transition states, and activation energy on potential energy diagrams for various mechanisms.
Solved Label the following multi-step reaction energy - Chegg Solved Label the following multi-step reaction energy | Chegg.com Science Chemistry Chemistry questions and answers Label the following multi-step reaction energy diagram AHn> 0 Reaction Intermediate Ealstep 2) Products Ea (step 1) Reactants Reaction progress
Chem 51A. Lecture 19. Organic Chemistry: Ch. 6. Energy Diagrams ... -11:11 Energy Diagram for Single Step Reaction-17:13 Transition State-26:06 Energy Diagram for Multi-Step Reaction-34:46 Steps in the Multi-Step Reaction-30:01 Reverse Reaction of Multi-Step Reaction-41:34 Rates of Reaction-47:15 Other Factors Affecting Rate. Required attribution ...
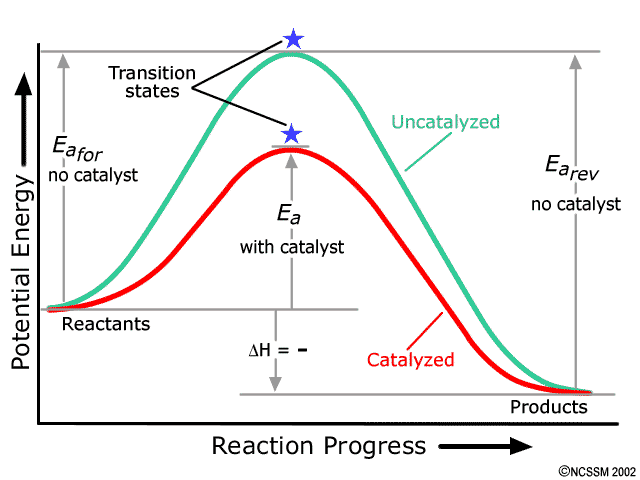
TIGER - NCSSM Distance Education and Extended Programs This diagram shows a potential energy diagram for a multi-step mechanism. NOplusH2_ver_1-640.gif This diagram shows a one-step mechanism for a reaction. NOplusH2_ver_2-640.gif This diagram shows a two-step mechanism for a reaction with the first step being rate determining. NOplusH2_ver_3-640.gif This diagram shows a two-step mechanism for a ...
Which step below is rate determining - Chemistry Stack Exchange 2 Answers. The second step is rate-determining. According to Wikipedia: Given a reaction coordinate (energy diagram), the rate determining step can be determined by taking the largest energy difference between any starting material or intermediate on the diagram and any transition state that comes after it. That transition state will then be ...
Multistep reaction energy profiles (video) | Khan Academy Many chemical reactions have mechanisms that consist of multiple elementary steps. The energy profile for a multistep reaction can be used to compare the activation energies of the different steps and identify the rate-determining step. The energy profile can also be used to determine the overall change in energy for the reaction.
Multi-Step Reaction - an overview | ScienceDirect Topics The multi-step reaction catalyzed by the PDH complex involves sequential reactions with the intermediates being relayed between the subunits via the flexible lipoyl domains. E1 catalyzes the decarboxylation of pyruvate forming 2- (1-hydroxyethyldiene)-TPP bound to E1 (TPP is thiamin pyrophosphate).
Mechanisms and Potential Energy Diagrams - Course Hero The potential energy diagram can illustrate the mechanism for a reaction by showing each elementary step of the reaction with distinct activation energy (see Figure below ). Figure 1. The potential energy diagram shows an activation energy peak for each of the elementary steps of the reaction.
Energy Diagrams of Reactions | Fiveable The energy diagrams below show what should be known for the test. Image Courtesy of Pinterest Before looking at the specifics of each, you should be aware of a few terms: PEreactants - The potential energy of the reactants PEproducts - The potential energy of the products Activation Energy - Energy necessary for the reaction to occur
Reaction Mechanisms and Multistep Reactions - Chemistry LibreTexts Multi-step (Consecutive) Reactions Mechanisms in which one elementary step is followed by another are very common. A+ B → Q B+ Q → C A+ 2B → C (As must always be the case, the net reaction is just the sum of its elementary steps.) In this example, the species Q is an intermediate, usually an unstable or highly reactive species.
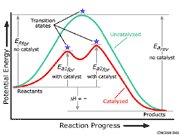

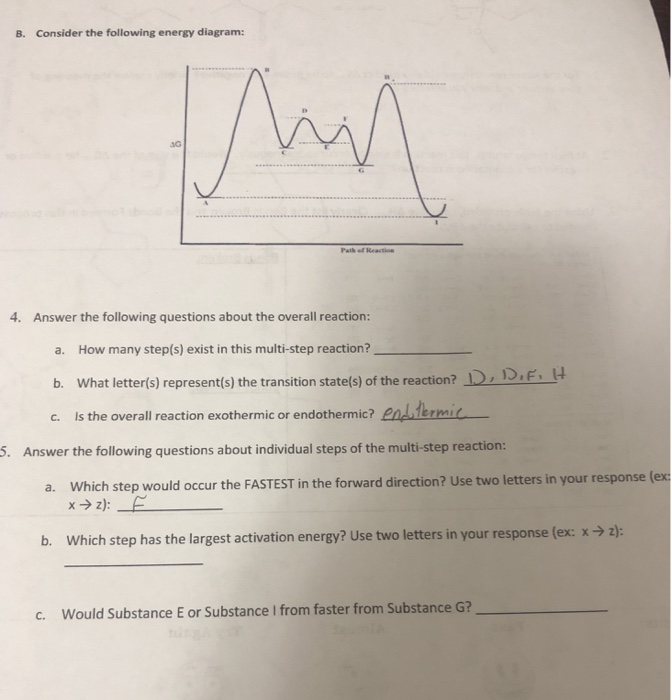

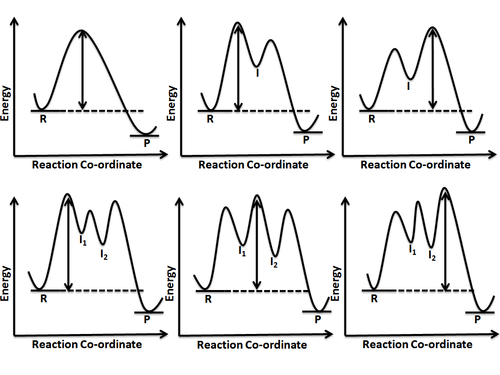


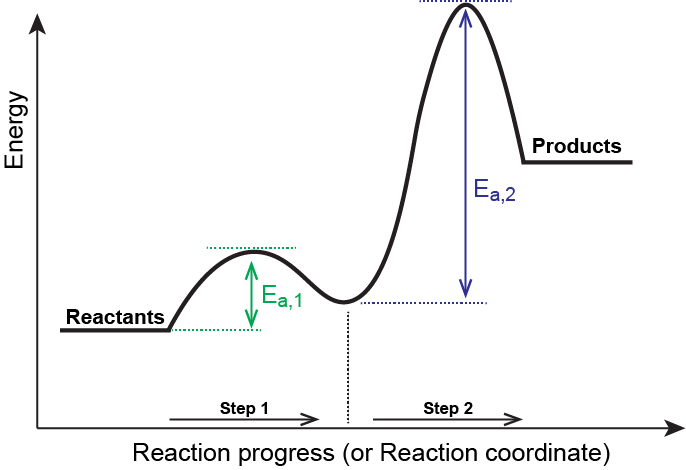














Post a Comment for "40 multi step reaction energy diagram"